Provide your information and you will get the link to the CDC Advancing HIV, STI and Viral Hepatitis Testing Conference for Avioq’s 2019 presentation.
VioOne HIV Profile Contact
Serologic assay for detection and differentiation of antibodies
directed to different gene products of HIV-1 and HIV-2
HIV Profile Intended Use
For in vitro Diagnostic Use
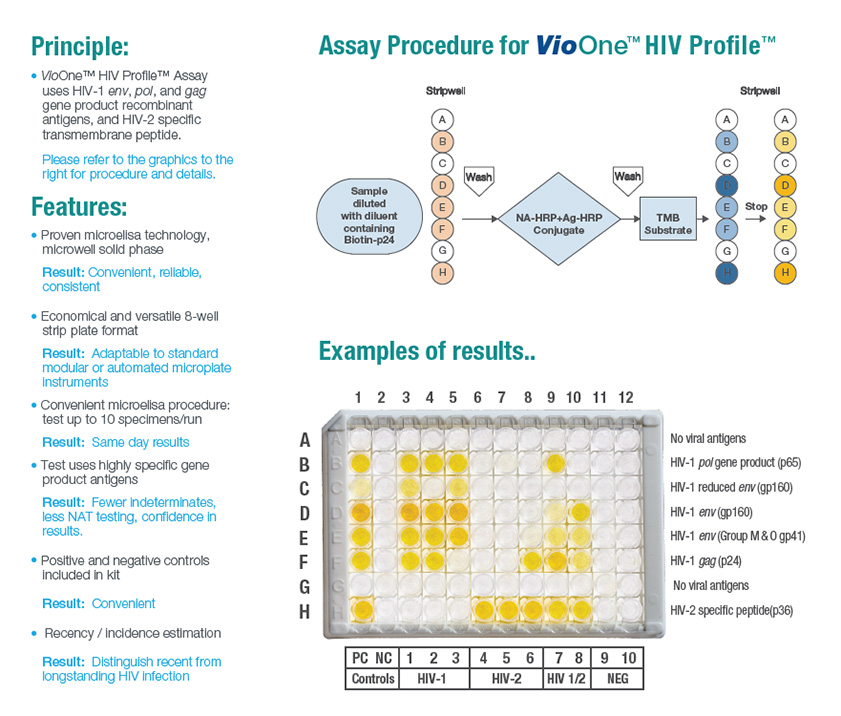
The VioOne™ HIV Profile™ Supplemental Assay is an enzyme-linked immunosorbent assay
(ELISA) for confirmation and differentiation of individual antibodies directed to various gene
products of HIV-1 (Group M & Group O) and HIV-2 in human serum or plasma. It is a more
specific test used to confirm the presence of HIV-1 and/or HIV-2 antibodies from specimens
that were repeatedly reactive in diagnostic screening procedures.
 Click here for the full brochure in PDF format
Click here for the full brochure in PDF formatFor In Vitro Diagnostic Use
Serologic assay for detection and differentiation of antibodies directed to
different gene products of HIV-1 and HIV-2
HIV Profile Intended Use
The VioOne™ HIV Profile™ Supplemental Assay is an enzyme-linked immunosorbent assay (ELISA) for
confirmation and differentiation of individual antibodies directed to various gene products of
HIV-1 (Group M & Group O) and HIV-2 in human serum or plasma. It is a more specific test used to
confirm the presence of HIV-1 and/or HIV-2 antibodies from specimens that
were repeatedly reactive in diagnostic screening procedures.
Avioq has recently received CE Mark for the new VioOne HIV Profile Supplemental Assay and is looking for European distributors in the IVD market place.
HIV Profile is a serologic assay for detection and differentiation of antibodies directed to different gene products of HIV-1 and HIV-2.
Please see our product page at VioOne HIV Profile Supplemental Assay Link.
Avioq has the experience, specialized facilities and mature Quality System to take your engineered mammalian and bacterial cell lines from the initial development phase to a high-throughput cGMP manufacturing process.
We optimize the production conditions for your cell line to ensure consistently high yields at the lowest production cost, and our design control procedures are capable of fully documenting the development process to support submission to Regulatory agencies.
Bio-manufacturing and analytical capabilities include recombinant protein production either in mammalian or bacterial cells, monoclonal antibodies produced in hybridomas, and viral lysates/virus produced from large-scale cell cultures.
Scalable purification processes based on standard or custom bioprocessing techniques available to enable large scale generation of biochemicals suitable for further development and/or licensing.
Avioq’s Design Control and Quality Systems are fully developed. Our facility has been inspected numerous times by FDA and ISO, as well as by external partners.
Avioq has met all FDA 21 CFR Part 820 and ISO 13485:2016 requirements for document and purchasing controls, complaint handling, CAPA, non-conformances, design/production/process controls, quality audits, risk management and training.
Serologic assay for detection and differentiation of antibodies
directed to different gene products of HIV-1 and HIV-2
Please Choose Your Region: